|
Two electrons in different orbitals will have a different. The Pauli exclusion principle states no two electrons (or other fermions) can have the identical quantum mechanical state in the same atom or molecule. (CC-BY-NC-SA Libretexts) (3) The Pauli exclusion principle states that it is impossible for two electrons of a multi-electron atom to have the same set of values for all four quantum numbers. If they are found in the same orbital, then it will result in a violation of Hund’s rule.Įxamples of elements having completely filled subshells are helium, neon, argon, and xenon. Hund's rule is that spin multiplicity must be maximized in the ground state. According to Hund’s rule, these two electrons will occupy separate orbitals with the same spin. It applies to any identical particles with half-integral intrinsic spinthat is, having s 1/2, 3/2. The Pauli exclusion principle is extremely powerful and very broadly applicable. The 1s and 2s sublevels are filled completely, and the 2p sublevel is filled partially. This statement is known as the Pauli exclusion principle, because it excludes electrons from being in the same state. According to this configuration, there will be two electrons in each of the 1s and 2s sublevels and 2 electrons in the 2p sublevel. What Is the Pauli Exclusion Principle The Pauli exclusion principle states that in a single atom, no two electrons will have an identical set or the same quantum numbers (n, l, m l, and m s ). 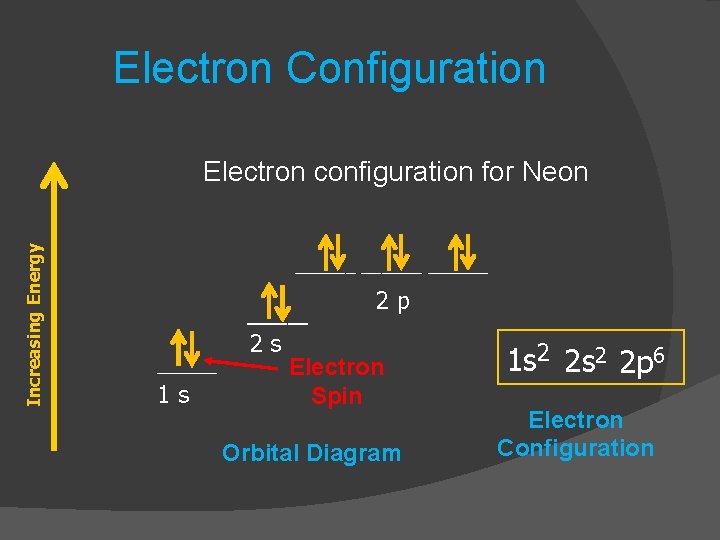
Then, a second electron with an opposite spin completes the occupancy.įor example, the electron configuration for a carbon atom is 1s 22s 22p 2. All the unpaired electrons have the same spin. The number of orbitals is as follows:Įach orbital is initially filled with one electron when the electrons fill them. The maximum number of electrons they can take is as follows:Įach sublevel is divided into orbitals, and each orbital can take a maximum of two electrons. The different sublevels are designated as s, p, d, and f. Such atoms do not have any empty orbital or unpaired electrons. The atom achieves a stable configuration when all the orbitals are filled. The Pauli exclusion principle specifies limits on how identical quantum numbers can be for two electrons in the same atom. The Pauli exclusion principle can be considered from two viewpoints. If the valence orbitals are not filled, the atom will be unstable and combine with another unstable atom to form chemical bonds. The modern state of the Pauli exclusion principle studies is discussed. quantum fields with half-integer spins can only be consistently quantized using Fermi-Dirac statistics (anticommutation relations), consequently they obey the exclusion principleīy "consistently quantized" I mean that the resulting free quantum field theory would satisfy the Wightman axioms, in particular, the energy spectrum would be bounded from below.Hund’s Rule and Electron Configuration Įlectron configuration can predict the stability of an atom.quantum fields with integer spins can only be consistently quantized using Bose-Einstein statistics (commutation relations), consequently they don't obey the exclusion principle.The result I'm referring to is called the spin-statistics theorem, it states that states that the position and momentum of an electron in an atom cannot be found precisely because measuring the electron changes its momentum. 
states that the position of an electron can be found by measuring its momentum. Now to how this is connected to the rotations. assumes that the electrons take positions predicted by Bohr's theory. 14) Write electron configurations for the following. This is exactly the Pauli exclusion principle: there couldn't be more than one particles filling the first-quantized state with momentum $p$. 13) Describe the Aufbau Principle, the Pauli Exclusion Principle, and Hunds Rule. We interpret these states as "there is no particle with momentum $p$" and "there is a particle with momentum $p$". The space of states free bosonic quantum field (Fock space) is constructed by applying the bosonic creation/annihilation operators 
The space of states free bosonic quantum field (Fock space) is constructed by applying the bosonic creation/annihilation operators. Pauli Exclusion Principle: no two electrons can be identified by the same set of quantum numbers (i.e. 
Hund's Rule: one electron goes into each until all of them are half full before pairing up. I am going to provide a sketch of the derivation here. 1 Answer Darshan Senthil Aufbau Principle: lower energy orbitals fill before higher energy orbitals. I am going to provide a sketch of the derivation here.įirst, consider the bosonic case. Pauli exclusion principle is a consequence of the Fermi statistics for free fermionic fields. Pauli exclusion principle is a consequence of the Fermi statistics for free fermionic fields.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
